CEFT - Infrastructure and Equipment


- SRU is the First Medical University in India having Animal facility (CEFT) certified for GLP compliance.
- 23,000 sq.ft spaces provides state-of-the art facilities for toxicology, genetic toxicology, pharmacology, behavioral, clinical & anatomic pathology evaluation.
- Access control by Biometric access system
- Double corridor systems falls under the Animal Bio Safety level II (ABSL II) category with 55:45 % air recycle ratio filtered through a 5 micron HEPA filter and 12 - 15 air exchanges per hour.
- Uninterrupted electric supply is ensured with two 1000 KVA generators.
- Environmental conditions (Temperature, Humidity, Pressure, Illumination and Noise levels) are centrally controlled, continuously monitored and recorded.
- Periodical third party analysis of feed, water and bedding material
- Exhaustive disaster management program to ensure the safety of the records, animals, and staff.
- Standard Pest control program, waste disposal and Fire safety regulations
- Separate rooms for quarantine, holding different species such as rabbits, guinea pigs, rats, mice and with individual rooms to conduct experiments.
- Individually ventilated caging system (IVC) for SPF and immune-compromised animals.
- Dedicated Non-GLP area for discovery and exploratory studies including Surgery suite and Behavioral procedures
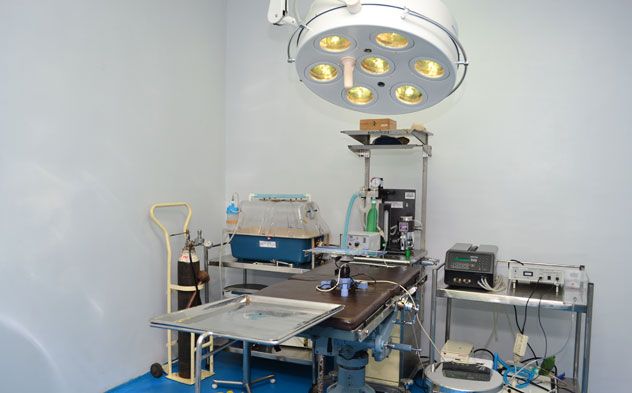
- Small Animal Surgical Theatre
- The suite is fitted with a ceiling mounted shadow less Operation theatre lamp with 70,000 lux capacity.
- Homoeothermic rodent operation board mounted on a hydraulic operation table.
- A dedicated inhalant anesthetic system.
- Head mounted operating cope, ICU, ECG and suction unit with all micro-surgicals.
- Stereotaxic apparatus for ICV injection
Major Units in CEFT - GLP
-
Quality Assurance Unit
- Direct Reporting & answerable only to Test facility Management
- Independent, dedicated & trained QAU personnel's
- Offers GLP training to all staffs
- Audits facility, process, & critical phases of the study
- Audits study plans for regulatory/GLP compliance
- Audits raw data, reports as per the GLP requirements
-
Test item control Office
TICO facility has discrete, well separated cabinets to receive and store test materials such as pharmaceuticals, crop protection products, and other chemicals. -
IT and documentation control Office
Well-structured Information and Technology division with validated computers and policies for high level assurance to centralized database system with RAID replication, security infrastructure to perform facility operations and disaster recovery backup. Units supports for Development &/or validation of the software programs, server management, archiving of e-data and back-up, System maintenance and SOP and Formats maintenance, updates and back-up. -
Archives – Wet & document
- Dedicated areas for dry and wet archives
- Fire proof sturdy door, fire and smoke detector and fire extinguisher
- Document archive has GI cabinet for storage of Documents and facility records,
-
Genetic Toxicology / cell culture / Bio-analytical
Equipped with- Western Blotting,
- PCR,
- Gel electrophoresis, Gel Documentation,
- ELISA plate reader, plate shaker,
- CO2 Incubator, BOD incubators,
- Biosafety cabinets (level II),
- Inverted Trinocular Microscope,
- Liquid Nitrogen containers etc.,
-
Pathology
Clinical Pathology- Fully automated hematology analyzer,
- Fully automated and semi-automated Biochemical Analyzer,
- Serum electrolyte analyzer,
- Urine analyzer,
- Automatic Hematocytometer,
- UV spectrophotometer,
- Cold storage units (up to -80ºC with GSM Data logger*),
- Incubators, Oven etc.
- Seasoned Histo-Technicians
- Automatic tissue processor- – Leica, & Spencer,
- Automatic tissue embedder,
- Microtomy – Leica, & Spencer,
- Automatic tissue Stainer
- Trinocular microscope with digital imaging
- Tissue Trimming as per Global Best practices endorsed by 'Society of toxicologic pathology'







